Spider Silk Unravelled: Structural Insights into Ampullate Spidroin via In Silico Modeling from the Cellar Spider, Crossopriza lyoni
Keywords:
Dragline silk, In silico, modeling, NT-AmSp, Spidroin, structureAbstract
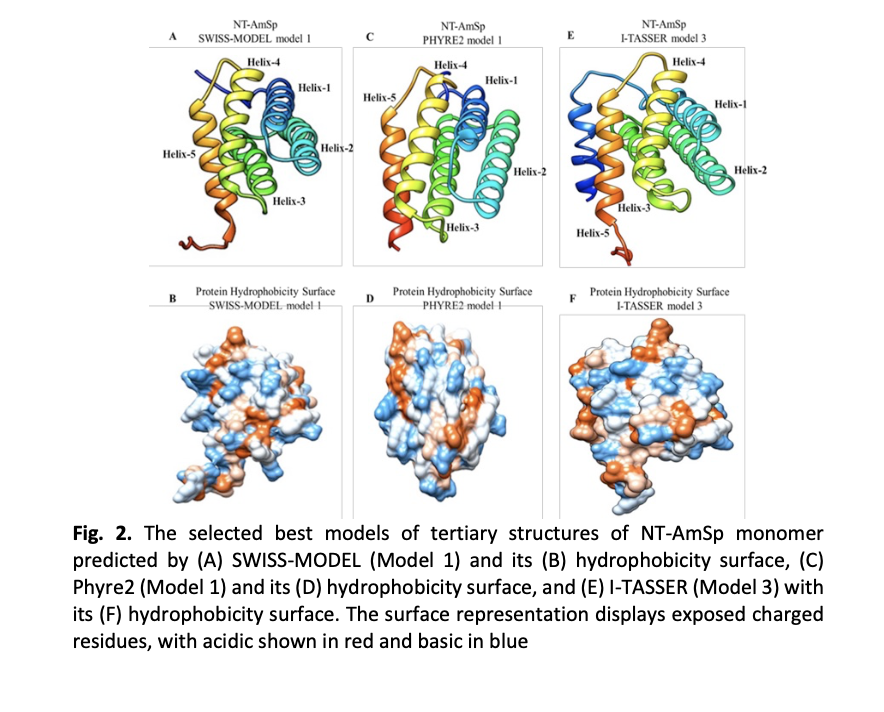
Spider dragline silk, or major ampullate silk, is a remarkably high-performing polymeric biomaterial with outstanding physical and mechanical properties due to its silk proteins, called spidroins. In particular, the N-terminus of spidroins plays a crucial role in silk fibre formation. Extensive research on the 3D N-terminal structure of major ampullate spidroins (MaSps) in orbicularian spiders has led to the neglect of major ampullate silks from non-orbicularians, despite their unique silk protein sequence, with a predominant focus on the adult stage rather than the early developmental stage. In this study, we elucidated the N-terminal ampullate spidroin (NT-AmSp) structure from the prenymph of the non-orbicularian species, Crossopriza lyoni. The NT-AmSp sequence of 155 amino acids was subjected to protein homology modeling, threading, and ab initio modeling through multiserver-based in silico predictions using SWISS-MODEL, Phyre2, and I-TASSER, respectively. The quality of each generated model was analysed using ProSA-web, QMEAN, and SAVES (parameters i.e., ERRAT, Verify3D, and Ramachandran plot) servers. Finally, the models were superimposed with an NMR-determined NT-MaSp from E. australis for similarity assessment using SuperPose. Models ranked first by both SWISS-MODEL and Phyre2 (Model 1) and Model 3 from I-TASSER with the highest C-score were chosen as the best predicted models. All models possessed five α-helices except for Model 3 with an additional α-helical conformation representing the signal peptide region. Overall, the models were of relatively good quality according to the analysis. The structure superimposition with E. australis NT-MaSp1 (4FBS) yielded an acceptable RMSD value between 2.0 Å and 3.0 Å. In silico structural modeling proves to be a powerful tool for assessing protein molecular functions. Elucidating the N-terminal structure of AmSp from C. lyoni prenymph may enhance our understanding of spidroin N-terminal diversity across spider taxa between adult and nymphal stages, and aid in developing soluble tags for heterologous protein expression.